For the Treatment of Cervical Radiculopathy
* Compared to posterior cervical fusion with lateral mass screws and rods.
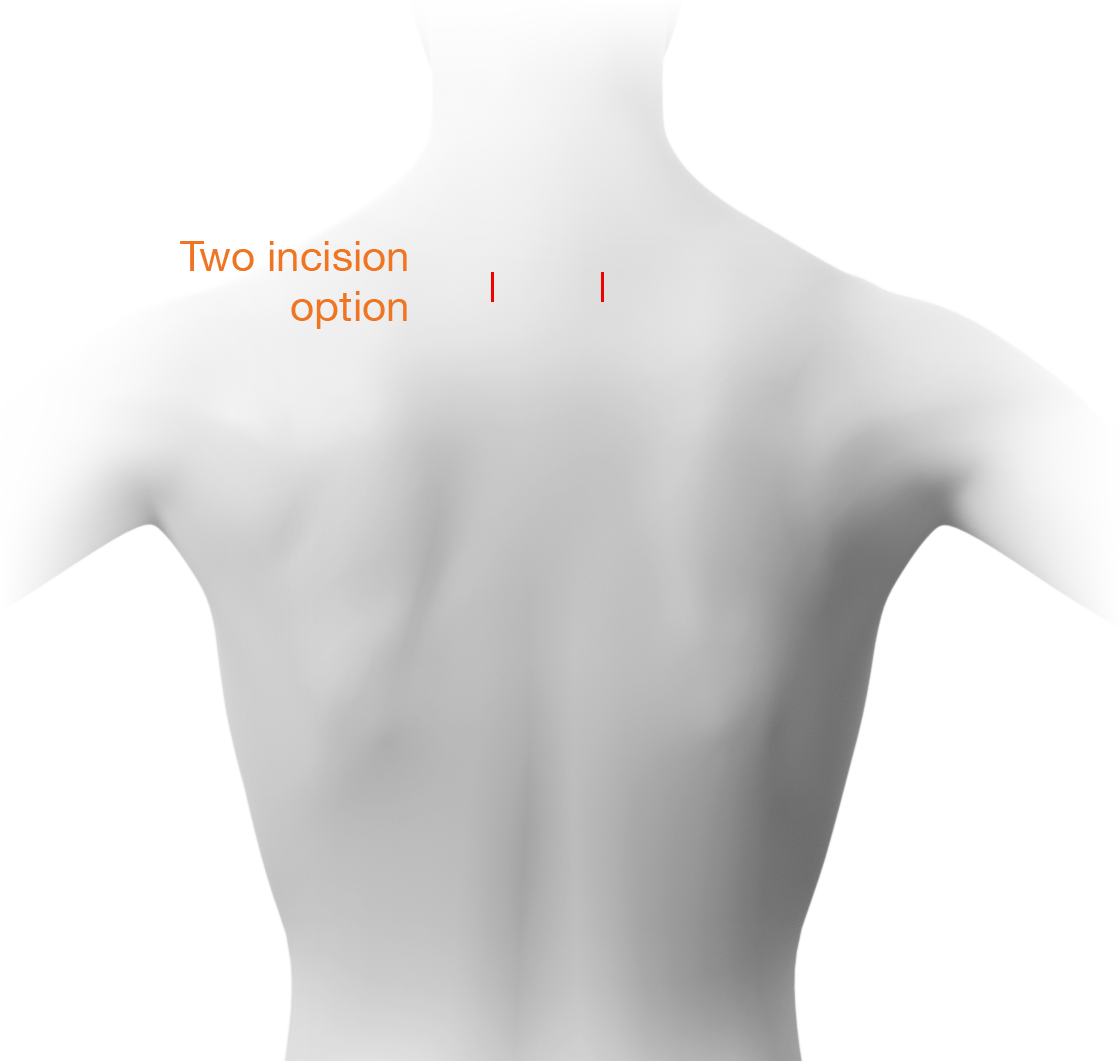

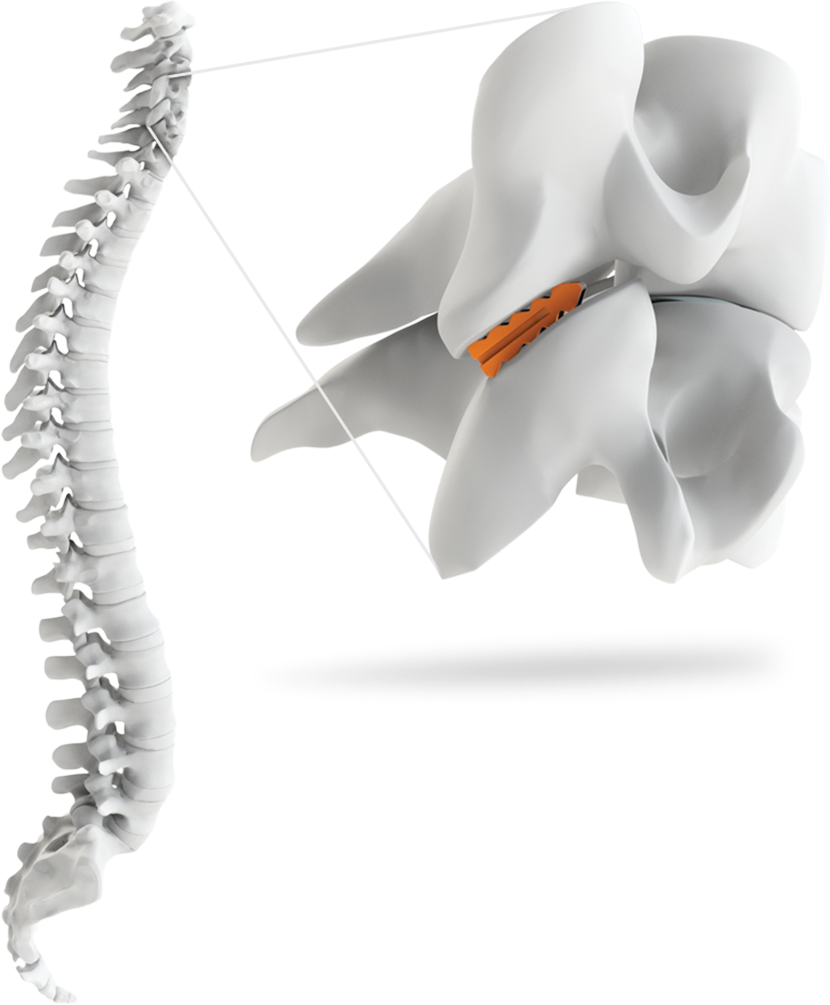

Indications
The GL-DTRAX® instruments, cages, and bone screws are indicated for use in skeletally mature patients for posterior cervical treatment at C3–C7 (inclusive) spinal levels for patients with single level radiculopathy due to degenerative disc disease (DDD) as defined by back pain of discogenic origin with degeneration of the disc confirmed by history and radiographic studies and/or degenerative disease of the facets.
Providence’s family of products offers a variety of surgical options to treat cervical radiculopathy. Patients should always consult their physician to determine which course of treatment is most appropriate.
Medical Disclaimer: Medical information or statements made within this site are not intended for use in or as a substitute for the diagnosis or treatment of any health or physical condition or as a substitute for a physician-patient relationship which has been established by an in-person evaluation of a patient. This website does not provide specific medical advice and does not endorse any medical or professional service or services obtained through information provided on this site or any links to or from this site.
Information made available through this website is not intended to replace the services of a physician or a health care professional acting under a physician Information made available through this website is not intended to replace the services of a physician or a health care professional acting under a physician’s supervision, nor does it constitute a doctor-patient relationship. Each individual’s treatment and/or results may vary based upon the circumstances, the patients’ specific situation, as well as the healthcare provider’s medical judgment and only after further discussion of the patient’s specific situation, goals, risks, benefits, and other factors.
Testimonials or statements made by any person(s) within this site are not intended to substitute for this discussion or evaluation or as a guarantee as to outcomes. Examples of treatment outcomes in this websites are not intended to convey and warranty, either express or implied, as to outcomes, promises, or benefits from treatment. Whether to accept any treatment by a patient should be assessed by the patient as to the risks and benefits of such procedures and only after consultation with a healthcare professional.
Before and after videos and photos published by Providence Medical Technology on this website are of actual patient results. These results are by no means an implied guarantee of individual outcomes. Individual outcomes vary according to the person and can be affected by many factors. Images and testimonials do not constitute a promise, guarantee, or representation of any particular outcome or experience. Use of providencemt.com constitutes acknowledgment and acceptance of the limitations and disclaimers described here. Use of this site constitutes acknowledgment and acceptance of these limitations and disclaimers. Further dissemination of this information in this site is prohibited without express written approval from Providence Medical Technology.
