March 19, 2026
Providence Medical Technology’s latest randomized controlled trial is designed to once again challenge the standard of care in spinal fusion surgery.
PLEASANTON, Calif., March 19, 2026 /PRNewswire/ -- Providence Medical Technology, Inc., a pioneer in less invasive solutions for spine surgery, today announced enrollment of the first patients in its prospective, randomized controlled trial (RCT) evaluating the CORUS-LX System for the treatment of degenerative lumbosacral disease. This multi-center RCT will enroll subjects indicated for two-level interbody fusion with supplemental percutaneous pedicle screw fixation and posterior fusion. Subjects will be randomized to receive posterior fusion either with the CORUS-LX System or with the surgeon’s standard posterior fusion technique.
Dr. Kade Huntsman of the Huntsman Spinal Clinic in Salt Lake City, Utah, was the first clinical investigator to treat an enrolled subject with the study device. The case utilized a circumferential fusion approach, integrating lumbar interbody fusion with percutaneous pedicle screws and the CORUS-LX System for fusion bed preparation and supplemental posterior stabilization and fusion. Huntsman joins Dr. Mohsin Fidai and Dr. Vivek Mohan as early-enrolling investigators in the study, which aims to evaluate subjects through 2027.
The Lumbar Blueprint: Building on Insights from the FUSE Study
This study is designed to replicate the rigorous clinical validation established by Providence’s FUSE study, which was the largest prospective RCT to evaluate three-level cervical fusion. Evidence from FUSE demonstrated that adding CORUS PCSS to an anterior cervical fusion delivers clinically superior composite fusion rates to anterior fusion alone. This new trial seeks to provide similar high-level insights for the lumbar spine.
“The FUSE Study was a turning point for cervical spine surgery because it clearly demonstrated that tissue-sparing, supplemental posterior fixation improves patient outcomes for 3-level patients,” said Scott Lynch, Chief Product Officer of Providence Medical Technology. “We believe this new trial will demonstrate that the CORUS-LX System provides similar biological and mechanical advantages for the lumbar spine.”
A Superiority Trial
In a medical device market often focused on “non-inferiority,” Providence is pursuing a statistical superiority endpoint. The trial aims to establish a new benchmark for two-level lumbar treatment options by comparing a treatment arm (CORUS-LX + Interbody Fusion + Pedicle Screws) against the current gold standard (Interbody Fusion + Pedicle Screws alone). The hypothesis is that the CORUS-LX construct will be a superior option for patients requiring L4-S1 lumbar fusion. The primary endpoint is to demonstrate superior 12-month composite fusion rates, with additional data collection on patient-reported outcomes and return-to-work measures through 24 months.
New Technology Addresses Inadequate Fusion Bed Preparation
Percutaneous pedicle screws are commonly used to stabilize the spine as a part of lumbar fusion surgery. While they offer a less invasive approach, percutaneous pedicle screws can limit a surgeon’s ability to “decorticate” or prepare the bone for a solid fusion. The inability to thoroughly prepare the fusion bed can lead to higher rates of pseudarthrosis (failure to fuse), particularly in high-risk patients. Conversely, the CORUS-LX System provides a tissue-sparing solution that enables thorough decortication and precise bone graft delivery while providing the stability of a traditional screw.
"Adding dedicated fusion implants in the facet joints provides more places for bones to fuse — both front and back of the spine," said Dr. Huntsman. "We expect clinical results observed in the neck to extend to the lower back, with faster recovery and fewer fusion failures."
Modernizing the Workflow: Precision via Surgical Navigation
A key component of the study involves using the CORUS-LX System with modern surgical workflows, specifically the surgical navigation systems from Medtronic and Globus Medical. By combining CORUS-LX with these leading navigation systems, surgeons can achieve high-precision placement of posterior implants through a tissue-sparing corridor.
About CORUS-LX
CORUS-LX is an integrated construct consisting of a stabilizer and two screws that is FDA-cleared for tissue-sparing posterior lumbar fusion and supplemental fixation. The system allows for extensive decortication and bone graft retention, serving as a dedicated fusion device that also provides significant stabilization.
About Providence Medical Technology, Inc.
Providence Medical Technology is a privately held medical device company focused on innovative solutions for cervical and lumbar spinal fusion. With a mission to improve clinical outcomes and reduce the economic burden of spine surgery, Providence is a leader in less-invasive, evidence-based surgical technologies. For more information, visit www.providencemt.com.
February 4, 2026
PLEASANTON, Calif., Feb. 4, 2026 /PRNewswire/ -- Providence Medical Technology (Providence), a pioneer in spinal fusion surgery technology, today announced the appointment of industry veteran Jim Hens as Chief Commercial Officer.
Hens has over 25 years of leadership experience in the orthopedic and spine industries and is joining the company to spearhead its commercial expansion and continue the company's industry-leading growth trajectory. He brings a proven track record of scaling medical device organizations, most recently as Managing Director of U.S. for Silony Spine, where he oversaw all U.S. operations. Before that, he was a General Manager at NuVasive (acquired by Globus Medical Inc. in 2023), where he managed its largest division for three years, achieving record growth.
Hens' appointment comes as Providence extends its rapid growth following publication of its landmark 202-patient FUSE Study in the journal Spine. The prospective, randomized controlled trial is the first study of its kind and demonstrates that multi-level Circumferential Cervical Fusion (CCF) — the fusion of all joints of the spinal segment — delivered clinically superior outcomes when compared to the more commonly performed Anterior Cervical Discectomy and Fusion (ACDF).1
"The company is at an exciting inflection point where Providence's clinical evidence is accelerating market demand," said Scott Lynch, Chief Product Officer for Providence. "Jim is a rare talent who combines deep operational expertise and spine industry knowledge with a visionary approach to sales leadership. His skill in successfully leading high-performance teams will be critical as we leverage the clinical superiority of our CORUS technology to make CCF the standard of care for cervical spine fusion patients, and in growing CORUS technology adoption in lumbar spine fusion patients."
"I am thrilled to join Providence," said Hens. "The data from the FUSE study is a watershed moment for the industry, proving that we can significantly reduce surgical failures through a tissue-sparing, circumferential approach. I look forward to leading this world-class team to ensure surgeons have the instruments, implants, and support they need to achieve significantly higher rates of fusion success for their patients."
Geared for Profitable Growth
In 2025, the Company achieved key milestones to accelerate growth, including rapid expansion of its direct sales force, the successful launch of its PCSS product line, and the introduction of a line of lumbar navigational compatible products to bring CORUS technology to lumbar spinal fusions. The company's focus on delivering proven patient outcomes through rigorous clinical research and truly differentiated product designs has driven 40% revenue growth in 2024 and 2025. In addition, Providence's unique business model delivered two consecutive years of positive Earnings Before Interest, Taxes, Depreciation, and Amortization (EBITDA) while investing significantly in clinical trials, new product innovation, and rapid expansion of its direct sales force.
About Providence Medical Technology, Inc.
Providence Medical Technology, Inc. is a privately held medical device company based in Pleasanton, CA. The company is dedicated to improving clinical outcomes for spinal fusion patients and preventing surgical failures through its tissue-sparing, clinically proven solutions, including its CORUS™ technology.
As with all medical devices, there are risks and considerations to device use. Please refer to the device labeling for a full discussion of potential risks, contraindications, warnings, precautions, and instructions for use. Rx only. For indications and safety information, visit: providencemt.com/safety
Reference:
1. Strenge KB, et al. Three-Level Anterior Cervical Discectomy and Fusion With or Without an Investigational Posterior Stabilization System Assessed Through 24 Months: A Multicenter Randomized Controlled Trial. Spine (Phila Pa 1976). 2025 Sep 1;50(17):1161-1170. PMID: 40331720.
SOURCE Providence Medical Technology, Inc.
Read MoreDecember 9, 2025
Pleasanton, CA – December 09, 2025 – Providence Medical Technology, Inc., an innovator in solutions for spinal surgery, announces that its CORUS™ Navigation Access System-GLX (“CORUS-GLX System”) is now compatible with Globus Medical's ExcelsiusGPS™ Robotic Navigation Platform for use during spinal fusion surgery. This new compatibility clearance strengthens Providence Medical Technology's lumbar fusion portfolio and underscores its commitment to improving surgical outcomes for high-risk spinal fusion patients.
The CORUS-GLX System is intended to assist surgeons in locating, accessing, and preparing facet joints for posterior fusion procedures in the lumbar spine (lower back). It enables surgeons to seamlessly integrate the CORUS instruments and implants with the ExcelsiusGPS™ Platform. Surgical guidance and robotic navigation are increasingly used in spinal procedures, providing enhanced visualization and precise control, particularly for less-invasive techniques.
The potential benefits of utilizing the CORUS-GLX System in combination with robotic navigation platforms include:
James Dowdell, MD, an Orthopedic Surgeon at Hospital for Special Surgery and Instructor of Orthopedic Surgery at Weill Cornell Medical College, commented, "Integrating the CORUS-GLX System positions spine surgeons to perform tissue-sparing posterior lumbar fusion and facet fixation using advanced surgical navigation. This compatibility provides a precise, reproducible workflow to achieve fusion of the entire spinal segment, which is vital for durable outcomes in high-risk lumbar fusion patients."
"We are thrilled to integrate our CORUS technology with another leading robotic navigation platform," said Jeff Smith, CEO of Providence Medical Technology. "This compatibility is another step forward in our mission to establish Total Segment Fusion (TSF) as the standard of care in spinal fusion. Surgeons who use navigation in lumbar spine surgery can now readily integrate our tissue-sparing facet fixation and posterior fusion technology into their surgical workflow."
For more information about the CORUS™ Navigation Access System-GLX, please visit providencemt.com.
About Providence Medical Technology, Inc.
Providence Medical Technology, Inc. is a privately held medical device company dedicated to improving clinical outcomes and preventing surgical failures for high-risk spinal fusion patients. The company has pioneered innovative, tissue-sparing solutions for both cervical and lumbar spine surgery, with a core mission to establish Total Segment Fusion (TSF) – fusion of all three spinal joints – as the new standard of care for spinal fusion patients. Providence's portfolio is supported by a robust body of clinical evidence, including randomized controlled trials. All instruments and implants are sterile-packaged and single-use to ensure consistent quality and performance.
Indications for Use: The CORUS™ Navigation Access System-GLX for use with the CORUS™ Spinal System is intended to be used during spinal surgery to assist the surgeon in locating and preparing facet joints in either open, or minimally invasive procedures. The CORUS™ Navigation Access System-GLX is specifically designed for use with the Globus Medical ExcelsiusGPS™ which is intended for use as an aid for precisely locating anatomical structures and for the spatial positioning and orientation of an instrument holder or guide tube to be used by surgeons for navigating and/or guiding compatible surgical instruments in open or percutaneous procedures provided that the required fiducial markers and rigid patient anatomy can be identified on CT scans or fluoroscopy.
As with all medical devices, there are risks and considerations to device use. Please refer to the device labeling for a full discussion of potential risks, contraindications, warnings, precautions, and instructions for use. Rx only. View product safety information: providencemt.com/safety
ExcelsiusGPS is a registered trademark of Globus Medical, Inc.
CORUS is a trademark of Providence Medical Technology, Inc.
MKT-PMT-790 Rev 0
August 14, 2025
PLEASANTON, Calif., Aug. 14, 2025 /PRNewswire/ -- Providence Medical Technology announces publication of initial FUSE study results in Spine, a leading journal for spine surgery. The journal presents peer-reviewed results of a pivotal, multicenter randomized controlled trial showing that tissue-sparing circumferential cervical fusion (CCF) significantly outperforms anterior cervical discectomy and fusion (ACDF) alone in treating three-level degenerative disc disease (DDD). The findings establish a new benchmark for the surgical treatment of multi-level cervical pathology.
The article titled "Three-level anterior cervical discectomy and fusion with or without an investigational posterior stabilization system assessed through 24 months: a multi-center randomized controlled trial" is published in the Sep 1 issue of Spine. This represents the first peer-reviewed results from the FUSE IDE trial, which enrolled 227 patients across 18 U.S. sites and compared outcomes between standard three-level ACDF and ACDF with Providence's CORUS™ Posterior Cervical Stabilization System (PCSS). Results demonstrated statistically significant improvements in the CCF arm in fusion success, safety, and the reduction of reoperation rates without adding procedural risk or recovery burden.
Key findings published in Spine include:
"The publication of this randomized controlled trial in Spine represents a significant advancement in our understanding of multi-level cervical fusion," said Dr. K. Brandon Strenge, Lead Author of the publication. "For the first time, we have Level I evidence demonstrating that supplementing 3-level ACDF with minimally invasive posterior stabilization and fusion not only substantially improves fusion rates, but also drastically reduces the need for subsequent revision surgeries. This rigorously conducted study, with its comprehensive fusion criteria and inclusive patient population, provides critical new clinical evidence that reshapes how we approach multi-level cervical fusion cases and how we improve outcomes in high-risk patients."
"The publication of the FUSE study in Spine represents a major milestone for the field of cervical spine surgery," said Jeff Smith, CEO of Providence Medical Technology. "The science is clear: tissue-sparing CCF with CORUS PCSS is safer, more effective, and more durable than ACDF alone in multilevel cervical fusion cases. As such, these results should prompt spine surgeons and payors alike to reconsider how we treat multilevel cervical DDD."
The CORUS PCSS system, now FDA-cleared for use in up to three cervical levels, is designed to achieve posterior fixation with minimal disruption to muscle and soft tissue. Its unique design enables stabilization across the facet joints through a posterior approach, avoiding the high morbidity associated with traditional open posterior fixation.
The FUSE study was conducted under FDA oversight as part of an Investigational Device Exemption (IDE) and formed the clinical basis for FDA clearance of the CORUS PCSS system in July 2024.
For more information about the FUSE study and to access the Spine publication, visit https://providencemt.com/fuse-study-spine-publication/.
About Providence Medical Technology
Providence is a privately held medical device company focused on tissue-sparing solutions for cervical spinal surgery. The company develops innovative spinal fusion systems designed to improve long-term outcomes and reduce the need for costly revision procedures.
MKT-PMT-771 Rev 0
SOURCE Providence Medical Technology, Inc.
Read MoreJuly 11, 2024
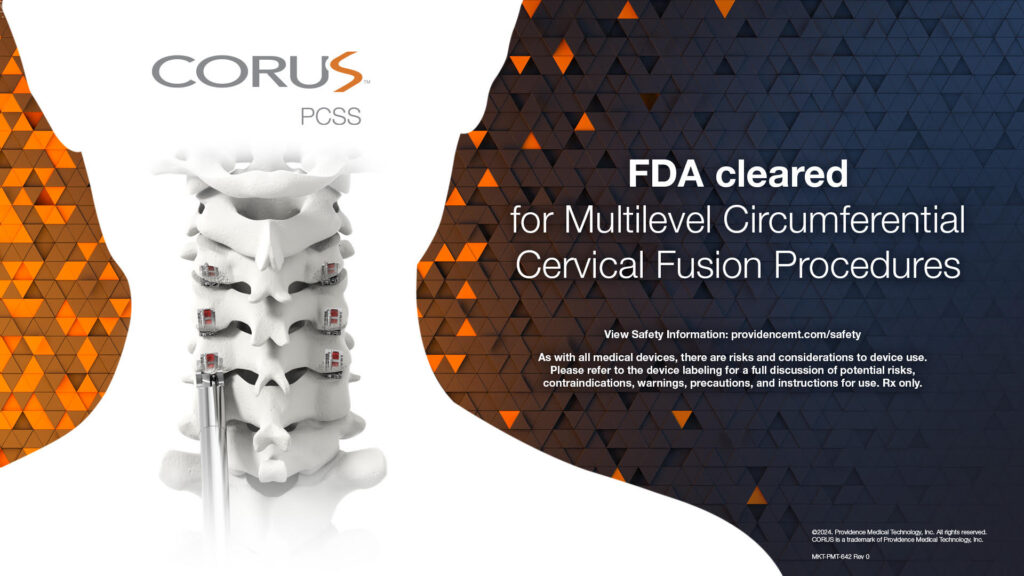
FDA clears CORUS™ PCSS for multilevel circumferential cervical fusion procedures based on results from the prospective, multicenter, randomized, controlled FUSE Study
PLEASANTON, Calif., July 11, 2024 /PRNewswire/ -- Providence Medical Technology announces FDA Clearance of its CORUS™ Posterior Cervical Stabilization System (PCSS) for the treatment of up to 3-level cervical Degenerative Disc Disease (DDD). The FDA clearance was based on results from the FUSE IDE study, a prospective, multicenter, randomized controlled trial comparing safety and effectiveness of Circumferential Cervical Fusion (CCF) versus Anterior Cervical Discectomy and Fusion (ACDF) alone in high-risk cervical fusion patients.
The FUSE Study was performed at 18 U.S. sites across 13 states and enrolled 227 patients aged 18 to 80, with symptomatic cervical DDD at three contiguous levels. The study randomized subjects to receive ACDF, the current standard of care, or CCF (ACDF plus posterior cervical fusion with the company's PCSS technology). Interim analysis was performed on over 200 subjects at 1 year follow-up and 100 patients at 2 years follow-up.
The interim analysis demonstrated the superiority of CCF in the study's primary endpoint of fusion success, highlighting the profound benefits of tissue-sparing CCF over ACDF alone:
Superior Fusion Success at 1 year: The study's primary endpoint demonstrated a 44.3% higher composite fusion rate of CCF over ACDF (61.0% vs 16.7%, p<0.001) using a strict definition of composite fusion. Fusion was deemed a success if all three consecutive levels demonstrated motion of less than 2 degrees on flexion-extension radiographs and continuous bridging bone was exhibited across the endplates of all three segments on thin-slice CT scans. Results were reported by an independent core imaging lab and verified by multiple radiologists.
Dramatic Difference in Revision Rates: Across all ACDF subjects followed through study completion, 22.8% (13/57) required subsequent surgical intervention (primarily due to symptomatic nonunion), compared to 1.7% (1/59) in the CCF arm.
Overall Safety Success at 2 years: The study's secondary endpoint measured overall safety success at 2 years using a composite of fusion success, lack of subsequent surgical interventions, maintenance or improvement in neurological success, and Neck Disability Index (NDI) improvement. The CCF arm exhibited a superior composite overall safety success rate at 24 months compared to the ACDF arm (CCF=50.8%, ACDF=22.8%, p<0.002).
No Increase in procedure-related Adverse Events: 3-level CCF with PCSS demonstrated statistically lower procedure-related adverse events than 3-level ACDF (p=0.005).
These findings underscore the transformative potential of CCF with the PCSS device in managing degenerative disc disease in high-risk cervical fusion patients. With minimal added operative morbidity, CCF delivers significant improvement to long-term outcomes over the current standard of care. As such, results from this pivotal FUSE study represent a consequential advancement that redefines the standard of care for patients with multilevel disease.
Dr. Pierce D. Nunley, MD, the Director of the Spine Institute of Louisiana and a Principal Investigator in the FUSE Study, commented on the significance of the findings, stating, "The results from this study provide very compelling evidence for the use of circumferential cervical fusion with PCSS over the standard ACDF treatment for multilevel cervical degenerative disc disease. CCF patients had significantly higher fusion rates and greater improvements in patient-reported symptoms and overall quality of life. Furthermore, the Secondary Surgical Intervention (SSI) rates demonstrate the durability of CCF with PCSS: only 1.7% of CCF subjects (1/59) who completed the study required SSI, compared to 22.8% of ACDF subjects (13/57). This study should cause spine surgeons and payors to re-examine their preferred approach to treating 3-level cervical DDD."
Jeff Smith, Chief Executive Officer of Providence Medical Technology, remarked, "The findings from the FUSE study mark a milestone in spinal surgery. These outcomes unequivocally demonstrate that 3-level CCF with CORUS PCSS has superior efficacy for spinal fusion and that 3-level ACDF fusion rates are unacceptably low. The high rates of 3-level ACDF failures and reoperation underscore how these patients need more to heal properly and achieve positive outcomes. The strength of this clinical evidence suggests that CCF with CORUS PCSS should become the new standard of care for multilevel fusion patients."
For more information about the FUSE study and its implications, please visit: www.providencemt.com
The FUSE Clinical Study aims to redefine the standard of care for this prevalent condition through rigorous scientific inquiry and innovative treatment approaches. https://clinicaltrials.gov/study/NCT04229017
Indications for Use:
CORUS™ Posterior Cervical Stabilization System (PCSS) is posterior spinal instrumentation with integrated screw fixation intended to provide immobilization and stabilization of spinal segments.
CORUS PCSS is placed through a posterior surgical approach in up to 3 consecutive levels of the cervical spine (C3-C7) and achieves bilateral facet fixation by spanning the facet interspace at each level with points of fixation at each end of the construct.
CORUS PCSS is intended as an adjunct to posterior cervical fusion (PCF) and is only intended to be used in combination with an anterior cervical discectomy and fusion (ACDF) at the same level(s).
CORUS PCSS is indicated for skeletally mature patients with degenerative disc disease (DDD). DDD is defined as radiculopathy and/or myelopathy, neck and/or arm pain of discogenic origin as confirmed by radiographic studies.
CORUS PCSS is to be used with autogenous bone and/or allogenic bone graft.
SOURCE Providence Medical Technology, Inc.
June 13, 2024
Pleasanton, CA – June 13, 2024 – Providence Medical Technology, Inc., an innovator in solutions for spinal surgery, announces that the U.S. Food and Drug Administration (FDA) has cleared the use of the CORUS™ Navigation Access System for use with the CORUS Spinal System during spinal surgery. The CORUS Navigation Access System is specifically designed for use with Medtronic's StealthStation™ System for posterior spinal fusion procedures. This FDA clearance strengthens Providence Medical Technology's portfolio and underscores its commitment to advancing spinal surgery for high-risk spinal fusion patients.
The CORUS™ Navigation Access System is intended to assist surgeons in locating, accessing, and preparing facet joints for posterior fusion procedures. It enables surgeons to utilize surgical navigation to perform procedures with its novel CORUS Spinal System. Surgical guidance and navigation have become increasingly utilized in spinal surgical procedures, particularly for less invasive surgical techniques.
The potential benefits of utilizing the CORUS™ Navigation Access System to perform a navigated CORUS fusion include:
Bryan Lee, MD, a neurosurgeon at Barrow Neurological Institute, commented, "The newly released CORUS navigation system offers simplicity and accuracy for placement of CAVUX® FFS-LX implants and provides the surgeon with a truly minimally invasive approach for posterior fusion, with a wide array of indications, including advanced disc degeneration and adjacent segment disease."
"We are thrilled to integrate our CORUS™ Spinal System with the leading surgical navigation platform and allow for incredible precision and safety in posterior fusion surgeries," said Jeff Smith, CEO of Providence Medical Technology. "This clearance signifies another leap forward in our mission to improve clinical outcomes for high-risk patients and prevent surgical failures of the spine.”
For more information about the CORUS™ Navigation Access System please visit providencemt.com.
As with all medical devices, there are risks and considerations to device use. Please refer to the device labeling for a full discussion of potential risks, contraindications, warnings, precautions, and instructions for use. Rx only. View product safety information: providencemt.com/safety
About Providence Medical Technology, Inc.
Providence Medical Technology, Inc. is a leading medical device company focused on advancing spine surgery technologies. Its commitment to improving clinical outcomes and reducing failures in high-risk spinal surgery has led PMT to develop a range of innovative solutions like CORUS™ Spinal Systems, CAVUX® Facet Fixation Systems, and ALLY® Bone Screws and Facet Screws.
Indications for use:
The CORUS™ Navigation Access System for use with the CORUS™ Spinal System is intended to be used during spinal surgery to assist the surgeon in locating and preparing facet joints in either open or minimally invasive procedures. The CORUS™ Navigation Access System is specifically designed for use with the Medtronic StealthStation™ System, which is indicated for any medical condition in which the use of stereotactic surgery may be appropriate, and where reference to a rigid anatomical structure, such as a long bone, or vertebra can be identified relative to a CT or MR-based model, fluoroscopy images, or digitized landmarks of the anatomy.
CORUS™ Spinal System-X is a set of instruments indicated to be used to perform posterior cervical fusion in patients with cervical degenerative disc disease.
CORUS™ Spinal System-LX is a set of instruments indicated to be used to perform posterior lumbar fusion in patients with lumbar degenerative disc disease.
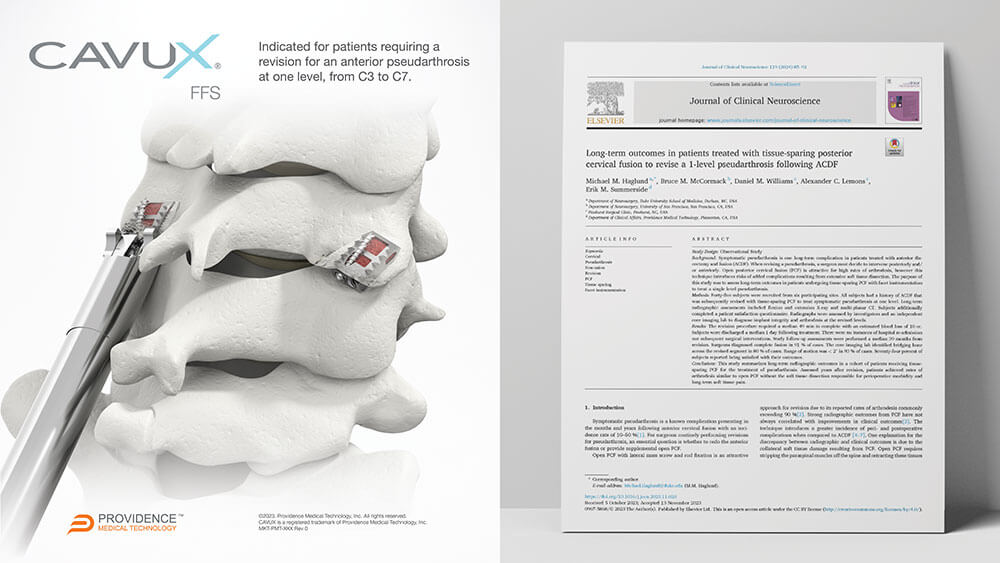
CAVUX® Facet Fixation System (CAVUX FFS) is an integrated construct comprised of a CAVUX Cage and a single ALLY Bone Screw. CAVUX FFS is placed bilaterally through a posterior surgical approach and spans the interspace with points of fixation at each end of the construct. CAVUX FFS is intended for temporary stabilization as an adjunct to posterior cervical fusion in skeletally mature patients. CAVUX FFS is indicated for patients requiring a revision for an anterior pseudarthrosis at one level, from C3 to C7, with autogenous and/or allogenic bone graft.
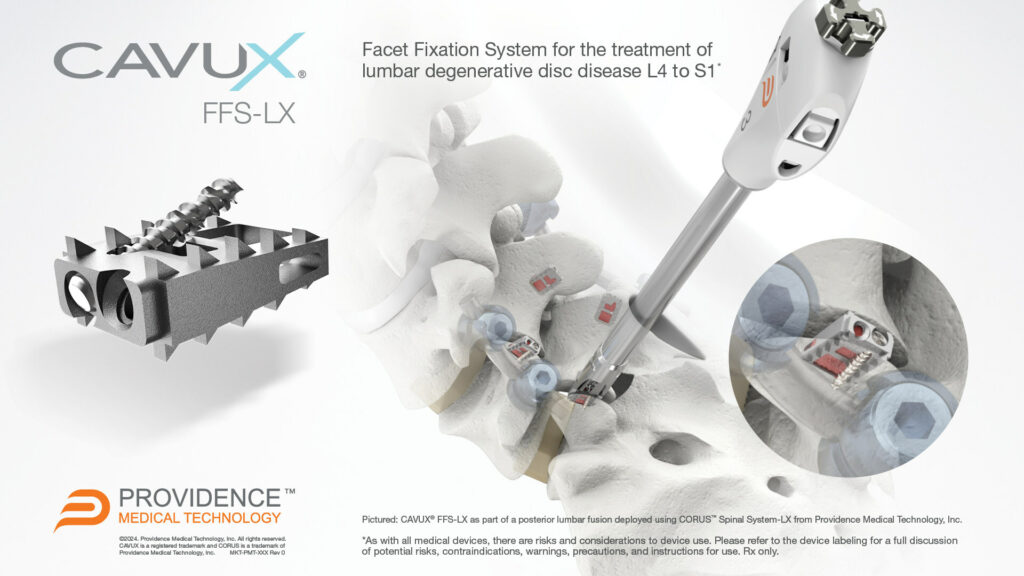
CAVUX Facet Fixation System, Lumbar (CAVUX FFS-LX) is an integrated construct comprised of a CAVUX Cage and an ALLY Bone Screw. CAVUX FFS-LX is placed bilaterally through a posterior surgical approach and spans the facet interspace with points of fixation at each end of the construct. CAVUX FFS-LX is intended to provide temporary stabilization as an adjunct to a 1 or 2 level interbody lumbar fusion with autogenous and/or allogenic bone graft and must be accompanied with an FDA-cleared intervertebral body fusion device implanted at the same spinal level(s), and may be used with a pedicle screw and rod system implanted at the same spinal level(s). CAVUX FFS-LX is indicated for the treatment of patients with lumbar degenerative disc disease (DDD) from L4 to S1 in skeletally mature patients who have failed conservative care.
SOURCE Providence Medical Technology, Inc.

January 10, 2024
CAVUX FFS-LX Achieved 96% Fusion Rate Verified by Independent Core Imaging Lab in Recent Clinical Study
Pleasanton CA, January 10, 2024 / PRNewswire/ -- Providence Medical Technology, Inc. (PMT), a medical device innovator focused on improving surgical outcomes for high-risk spine surgery patients, announced that the U.S. Food and Drug Administration (FDA) has cleared its CAVUX® FFS-LX: Lumbar Facet Fixation System for use in lumbar spinal fusion surgery.
CAVUX® FFS-LX is a novel integrated cage and screw system that is implanted bilaterally in the facet joints to treat lumbar degenerative disc disease (DDD). The implant spans the facet interspace with points of fixation at each end of the construct to offer additional stabilization for 1- or 2-level lumbar interbody fusion. CAVUX® FFS-LX may be used with or without pedicle screws and rods and is implanted using the company’s CORUS™ Spinal System-LX tissue-sparing access and spinal fusion system.
The clearance marks the company's expansion into the $2 billion lumbar spine market characterized by a higher surgical failure rate than in the cervical spine. Patients who fail to fuse after a lumbar spine fusion surgery face a substantial risk of added complications, suffering, and costly revision procedures. CAVUX® FFS-LX was designed to offer increased stabilization following lumbar fusion procedures in order to increase fusion rates and reduce future complications and reoperations.
The clearance was supported by clinical study data demonstrating a strong safety and efficacy profile. Summary clinical study data reviewed by the FDA in considering the clearance included:
CAVUX® FFS-LX (Facet Fixation System, Lumbar) is a novel integrated cage and screw system to treat lumbar degenerative disc disease that is now FDA-cleared for use in lumbar spinal fusion surgery.
December 7, 2023
Latest Study Published in Journal of Clinical Neurosurgery adds to Robust Body of Evidence Supporting Tissue-Sparing Posterior Cervical Fusion to Treat High-Risk Cervical Fusion Patients
PLEASANTON, Calif., Dec. 7, 2023 /PRNewswire/ -- Providence Medical Technology, Inc., a medical device innovator focused on improving surgical outcomes for high-risk spine surgery patients, today announced a new publication in the Journal of Clinical Neuroscience. Dr. Michael M. Haglund and colleagues authored the publication on the long-term outcomes of patients undergoing tissue-sparing Posterior Cervical Fusion (PCF) to revise a 1-level pseudarthrosis following a failed Anterior Cervical Discectomy and Fusion (ACDF). View Release
May 11, 2023
PLEASANTON, Calif., May 11, 2023 /PRNewswire/ -- Providence Medical Technology, Inc. (PMT), a leader in medical device innovation for spine surgery, today announced the completion of enrollment in its FUSE Study — a prospective, multicenter, randomized, Investigational Device Exemption (IDE) clinical study evaluating the safety and effectiveness of its Posterior Cervical Stabilization System (PCSS) in 3-level cervical fusion patients. The study enrolled approximately 230 patients across 18 leading spinal surgery sites in the United States to evaluate PCSS in combination with Anterior Cervical Discectomy and Fusion (ACDF) compared to ACDF alone in a superiority study.
The study began in May 2020 and is designed to evaluate Circumferential Cervical Fusion (CCF) in high-risk cervical fusion patients by producing level-1 clinical evidence. The study includes patients at risk for failure of a standard ACDF fusion procedure due to risk factors such as 3-level disease, smoking, advanced age, and diabetes. While a traditional ACDF procedure is generally associated with positive outcomes in otherwise healthy patients, the success rate for these high-risk patients is significantly less favorable. Of the over 300,000 anterior cervical fusion procedures performed annually, roughly 30% involve patients with significant risk factors for nonunion. This study is investigating whether these patients could benefit from a tissue-sparing posterior cervical fusion as an adjunct to the ACDF.
The first comparative analysis for fusion and other clinical outcomes will occur when 100 patients reach two years of follow-up. The 100th patient is scheduled to return for their two-year follow-up visit at the end of 2023. View Release
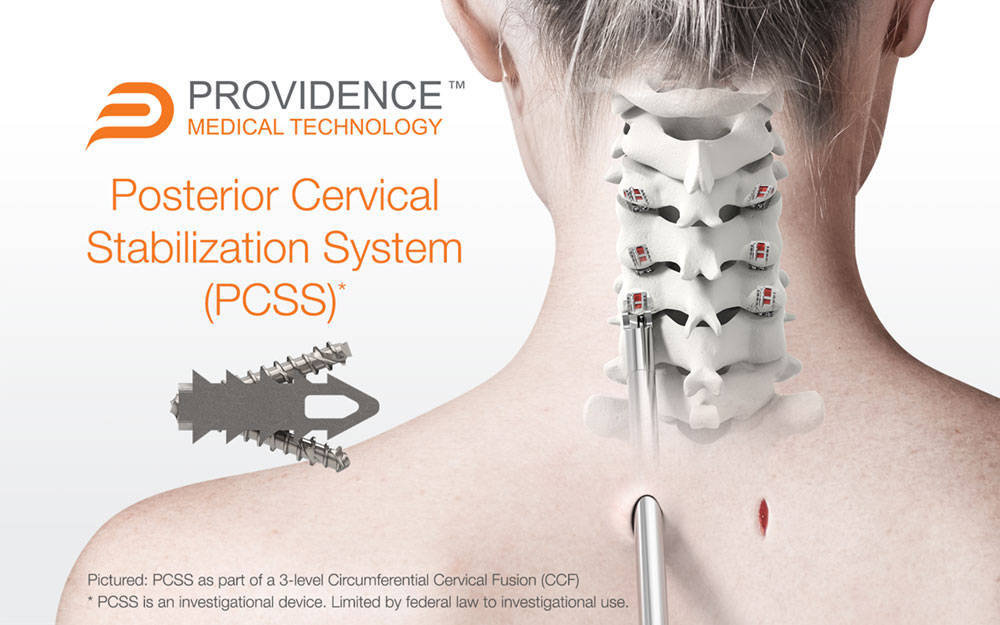
PCSS is an investigational implant system composed of non-segmental instrumentation with integrated screw fixation designed to provide immobilization and stabilization of spinal segments. PCSS is designed to achieve bilateral facet fixation at each level by spanning the interspace with points of fixation at each end of the construct.
December 15, 2022
Providence Medical Technology, Inc., a medical device innovator focused on improving surgical outcomes for high-risk spine surgery patients, announced that the U.S. Food and Drug Administration (FDA) has cleared its CAVUX® Facet Fixation System (FFS) for a new indication. The Facet Fixation System (FFS) is a novel integrated cage and screw system that is implanted bilaterally in the facet joints to provide stabilization until fusion occurs. View Release
Read More