

Book a 15-minute introductory call with our VP of Clinical Affairs, Matt Jenkins
Connect with Matt Jenkins, our VP of Clinical Affairs, to see if this study is the right fit.
Not Ready to Connect?
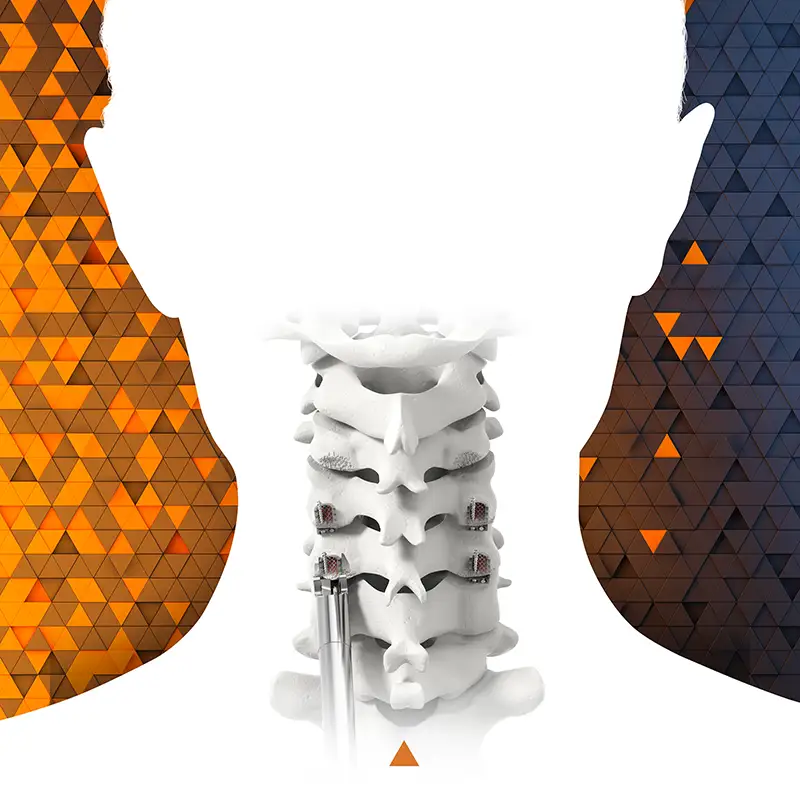
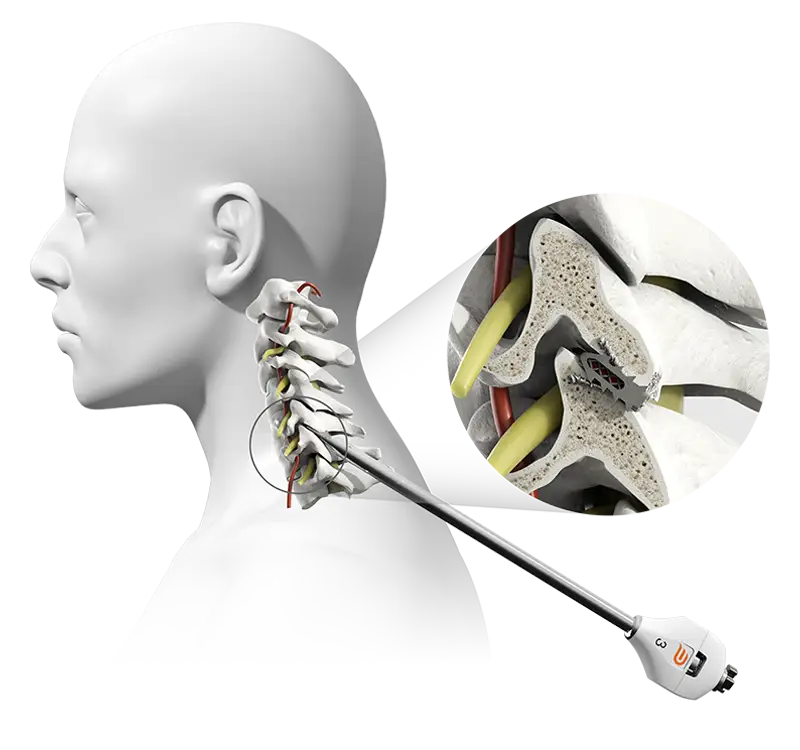
We invite you to learn more about the technology under evaluation via our Quick Study Video
